

Total cross sections for the ionization and excitation of atoms and molecules by electron impact is one of the essential sets of data needed in a wide range of applications, such as modeling plasmas for plasma processing of semiconductors, designing mercury-free fluorescent lamps, assessing the efficiency of ion gauges, normalizing mass spectrometer output, diagnosing plasmas in magnetic fusion devices, and modeling radiation effects on materials.
In addition to total ionization cross sections, sometimes differential
ionization cross sections are needed. We present below total ionization cross
sections for a large numbers of atoms/molecules, and singly differential
ionization cross sections (energy distribution of ejected electrons) for H, He,
and H2. Excitation cross sections for H, He, and Li are also
presented.
The theory used in this ionization cross section database is specifically
designed for electron-impact ionization. It is versatile and can provide cross
sections for atoms as well as molecules. The theory, referred to as the
Binary-Encounter-Bethe (BEB) model [1], combines
the Mott cross section with the high-T behavior of the Bethe cross
section. The theory does not use any fitting parameters, and provides a simple
analytic formula for the ionization cross section per atomic/molecular orbital.
The total ionization cross section for a target is obtained by summing these
orbital cross sections. Four orbital constants--the binding energy B,
the orbital kinetic energy U, the electron occupation number
N, and a dipole constant Q--are needed for each orbital, and
the first three constants are readily available from the ground-state wave
function of the target atom or molecule. The basic formula for the ionization
cross section per orbital is [1]:
|
|
(1) |
where t = T/B, u = U/B,
|
|
(2) |
When df/dW is unknown, one can put Q = 1 as a further approximation. The constant n on the right-hand side (RHS) of Eq. (1) is used for ion targets and valence orbitals of large atoms as indicated at the end of this section. Unless noted otherwise, use n = 1.
The BEB cross section is not very sensitive to the accuracy of the orbital constants used except for the value of the lowest B. A vertical ionization energy is recommended for the lowest B. We used an experimental value if it is known for a target. Otherwise theoretical values were used. The B values marked "OVGF" were calculated using the outer-valence Green's function method, those marked "CCSD" using the coupled cluster single double excitations, and those marked "CCSD(T)" using the CCSD plus triple excitations included perturbatively. Orbital constants obtained from the Hartree-Fock or similar wave functions are adequate. The resulting cross sections are accurate to 5 % to 20 % from threshold to T ~ 1 keV in most targets presented in this database.
As was demonstrated in a series of publications [1-11] the BEB model (with Q = 1 in most cases) was found to reproduce known ionization cross sections accurately for small atoms and a variety of large and small molecules from H2 to SF6. Also, the BEB model works well for radicals as well as stable molecules. In many cases, the theory agrees with experiments in peak values within 10 %. Unlike most theories, the BEB model is reliable near the threshold as well.
Production of a doubly charged ion or two singly-charged molecular fragments resulting from inner-shell ionization can be estimated by doubling the ionization cross sections of individual atomic/molecular orbitals whose binding energies B exceed the double ionization energy (approximately B > 40 eV) [6]. Inclusion of such contributions in the BEB theory is indicated by "Yes" in the atomic/molecular orbital constant column marked "DblIon," e.g., for C2F6 and C3F8 [7].
Through this WWW presentation, a user can (a) download orbital constants and ionization cross sections for an atom or molecule of interest as a text (ASCII file), (b) calculate the total ionization cross section at a given incident energy T online, (c) look at the graph for an atom or molecule with comparisons between the BEB model and experiments (data points for experiments, curves for theory unless otherwise specified), (d) zoom into a segment of the BEB cross section between specific incident energy limits, e.g., 5 eV to 100 eV, or (e) calculate the energy distribution of ejected electrons for H, He, and H2 online.
The molecules table and atoms table provide access to the data for those molecules and atoms to which the BEB model has been applied.
This database is updated frequently by adding new atoms and molecules as their cross sections become available. The BEB model is a simplified version of the BED model described in Section B below.
For singly-charged molecular ions, n = 2 is used on the RHS of Eq. (1) [Example: H2+]. Also, if the Mulliken population analysis of a valence molecular orbital indicates the leading component (> 50 %) consists of a specific atomic orbital with the principal quantum number n > 2, then this value of n is used in Eq. (1) [Example: CS2].
For atoms, if the principal quantum number (pqn) of an orbital is 3 or greater,
then use n = pqn in Eq. (1). If pqn is 1 or 2, then use
n = 1.
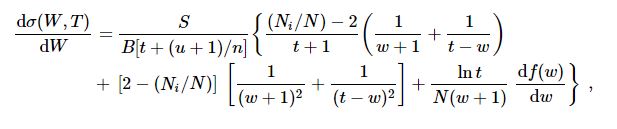 |
(3) |
where w = W/B, df(w)/dw = the
continuum dipole oscillator strength for ejecting an electron of kinetic energy
W by photoionization, and ![]() (df/dw)dw.
(df/dw)dw.
| (4) |
and the values of the coefficients a, b, ... and the corresponding SDCS are presented in the web pages of individual targets for T and W values requested by the user, or a table of SDCS for a T value and preselected values of W. The resulting dσ/dW is normalized to match the total ionization cross section. Sample comparisons between experimental data and theory for SDCS [9] are available online.
As is true for any theory, the BEB model for both total and differential
cross sections has its limits. For instance, the BEB model presented here
is a nonrelativistic theory, and therefore should not be used for
T > 10 keV. For a relativistic extension of the
BED/BEB model see [12].
| (5) |
where E is the excitation energy.
Scaling with just the excitation energy E, called E scaling, converts Coulomb Born cross sections (σ CB) for electron-impact excitation of singly charged atomic ions to highly accurate cross sections at all incident energies T, although the original Coulomb Born cross sections are reliable only at high T [14]. The E scaled cross section σE is defined as:
| (6) |
The most significant source for indirect ionization is the excitation of an inner-shell electron to an excited valence state (e.g., 3s → 3p excitation in aluminum), which is an unstable excited state. This excited state can decay by either emitting a photon or by ejecting an electron. The latter is called excitation-autoionization (EA). When the energy level of the excited state is not very high compared to the lowest ionization energy, EA dominates. For the ionization cross sections of atoms in this database, cross sections for significant EA (usually electric-dipole and spin allowed excitations) have been calculated using the BE and E scalings discussed in Section C above, and the EA cross sections are included in the total ionization cross sections.
For further information, please contact:
Dr. Karl Irikura
Chemical and Biochemical Reference Data Division
National Institute of Standards and Technology
Gaithersburg, MD 20899-8320
phone 301-975-2510; fax 301-869-4020
email Feedback.
