The electric dipole transitions are given by the following selection rules:
| (eq20) |
where a molecule-fixed cartesian coordinate system, with the z-axis
along the molecular axis, is employed. The operators, Lz,
L+, and L_ are the three spherical components of the
electronic orbital angular momentum; Sz, S+,
and S_ are the equivalent operators for electron spin and
Jz, J+, J_ for the total angular
momentum. The parameters B, A, and γ are functions of the internuclear distance and, thus, may be
defined in terms of a power series in ![]() as:
as:
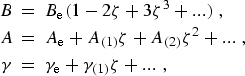 |
(eq20a) |
The eigenvalue solution of the Hamiltonian above is normally achieved by a perturbation method which takes into account the mixing of various vibrational states, and the mixing of various electronic states with the ground state. In this way centrifugal distortion terms, the vibrational dependence of the molecular parameters, l-uncoupling and Λ- or Ω-doubling can be determined.
There are a variety of possible approximations employed to describe the
observed microwave spectra. The method used depends on how close the angular
momenta coupling in a specific: molecule corresponds to Hund's coupling case
(a). Formulations employed for intermediate coupling cases, like that
for OH and NO, are given in ref. [13] and
[14]. The determinable parameters are
The appropriate formulation for coupling cases close to Hund's case (a),
e.g., ClO and NS, are given in ref. [16]. The
determinable parameters are
The rotational constant
The hyperfine coupling Hamiltonian given in ref. [10]
is evaluated in ref. [14] to first order for the
magnetic and nuclear electric quadrupole interactions. Although the first order
perturbation treatment is adequate for the interpretation of the microwave
spectra, the more detailed analysis in ref. [12] is
necessary to adequately describe the radiofrequency spectrum of NO. The
determinable parameters are the magnetic coupling constants a, b,
c, and d, as well as the quadrupole coupling constant, eQq,
which is proportional to the electric field gradient at the nucleus in the
direction of the molecular axis, and ![]() , which is proportional to the
field gradient perpendicular to the molecular axis. In molecules with coupling
cases close to case (a), the determinable parameters are functions of
combinations of the constants a, b, c, and d.
, which is proportional to the
field gradient perpendicular to the molecular axis. In molecules with coupling
cases close to case (a), the determinable parameters are functions of
combinations of the constants a, b, c, and d.
| Effective rotational constants in the | ||
| Centrifugal distortion correction constants in the | ||
| Ω-doubling parameters, | ||
|
|
(eq21) | |
|
|
(eq22) | |
| peff | Λ-type doubling
constant in the | |
| Magnetic hyperfine coupling constants (MHz) where, | ||
|
|
(eq23) | |
|
|
(eq24) | |
|
|
(eq25) | |
|
|
(eq26) | |
| Here μB is the Bohr magneton, μN is the nuclear magneton, and gN is the nuclear g-value. | ||
| eQq | Quadrupole coupling constant along the
molecular axis, where |
|
| Quadrupole coupling constant perpendicular to
the molecular axis, where |
||
| A | Spin-orbit coupling constant defined by the power series,
expansion, | |
| γ | Spin-rotation coupling constant defined by the power series
|
|
|
|||