
| Electric dipole (E1) ("allowed") |
Magnetic dipole (M1) ("forbidden") |
Electric quadrupole (E2) ("forbidden") |
||
|---|---|---|---|---|
| Rigorous rules | 1. | Δ J = 0, � 1 (except 0 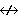 0) 0) |
Δ J = 0, � 1 (except 0 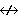 0) 0) |
Δ J = 0, � 1, � 2 (except 0 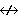 0, 0,1/2 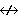 1/2, 0 1/2, 0 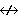 1) 1) |
| 2. | ΔM = 0, � 1 (except 0 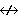 0 0when Δ J = 0) |
ΔM = 0, � 1 (except 0 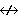 0 0when Δ J = 0) |
ΔM = 0, � 1, � 2 | |
| 3. | Parity change | No parity change | No parity change |
|
| With negligible configuration interaction |
4. | One electron jumping, with Δl = � 1, Δn arbitrary |
No change in electron configuration; i.e., for all electrons, Δl = 0, Δn = 0 |
No change in electron configuration; or one electron jumping with Δl = 0, � 2, Δn arbitrary |
| For LS coupling only |
5. | ΔS = 0 | ΔS = 0 | ΔS = 0 |
| 6. | ΔL = 0, � 1 (except 0 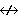 0) 0) |
ΔL = 0 Δ J = � 1 |
ΔL = 0, � 1, � 2 (except 0 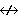 0,
0 0,
0 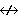 1) 1) |
|
|
|
(13) |
where Aki is the atomic transition probability and
Nk the number per unit volume (number density) of excited
atoms in the upper (initial) level k. For a homogeneous light source of
length l and for the optically thin case, where all radiation escapes,
the total emitted line intensity (SI quantity: radiance) is
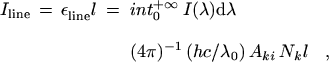 |
(14) |
where I(λ) is the specific intensity at wavelength λ, and λ0 the wavelength at line center.
|
|
(15) |
is used, where I(λ) is the incident intensity at wavelength &ambda;, e.g., from a source providing a continuous background, and I′(λ) the intensity after passage through the absorbing medium. The reduced line intensity from a homogeneous and optically thin absorbing medium of length l follows as
|
|
(16) |
|
|
(17) |
| |
(18) |
where ψi and ψk are the initial- and final-state wave functions and Rik is the transition matrix element of the appropriate multipole operator P (Rik involves an integration over spatial and spin coordinates of all N electrons of the atom or ion).
|
|
(19) |
Numerically, in customary units (A in s-1, λ in �,
S in atomic units),
|
|
(20) |
and for S and ΔE in atomic
units,
|
|
(21) |
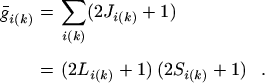 |
(22) |
The Aki values for strong lines of selected elements are given. For comprehensive numerical tables of A, f, and S, including forbidden lines (see Sources of Spectroscopic Data).
Experimental and theoretical methods to determine A, f, or S values as well as atomic lifetimes are discussed in Atomic, Molecular, & Optical Physics Handbook, Chaps. 17, 18, and 21, ed. by G.W.F. Drake (AIP, Woodbury, NY, 1996).
| SI units a | Numerically, in customary units b | |||
|---|---|---|---|---|
| Electric quadrupole | ||||
| Magnetic dipole | ||||
| a | A in s-1, λ in m. Electric quadrupole: S in m4 C2. Magnetic dipole: S in J2 T-2. |
| b | A in s-1, λ in �. S in atomic units:
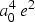 = 2.013 � 10-79
m4 C2 (electric quadrupole), = 2.013 � 10-79
m4 C2 (electric quadrupole),
|
Oscillator strengths f are not used for forbidden transitions, i.e., magnetic dipole (M1), electric quadrupole (E2), etc.
[Numerical example: For the
1s2p 1P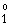 - 1s3d 1D2
(allowed) transition in He I at 6678.15 �:
gi = 3; gk = 5;
Aki = 6.38 � 107 s-1;
fik = 0.711;
S = 46.9 a02 e2.]
- 1s3d 1D2
(allowed) transition in He I at 6678.15 �:
gi = 3; gk = 5;
Aki = 6.38 � 107 s-1;
fik = 0.711;
S = 46.9 a02 e2.]
|
|
(23) |
| |
(24) |
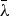 is the weighted
("multiplet") wavelength in vacuum:
is the weighted
("multiplet") wavelength in vacuum:
|
|
(25) |
|
|
(26) |