 |
#1
 General General |
Only units of the SI and those units recognized for use with the
SI are used to express the values of quantities. Equivalent values
in other units are given in parentheses following values in acceptable
units only when deemed necessary for the intended audience. |
 |
#2
 Abbreviations Abbreviations |
Abbreviations such as sec, cc, or mps are avoided and only standard
unit symbols, prefix symbols, unit names, and prefix names are
used. |
| proper: |
s or second; cm3 or cubic centimeter; m/s or meter per second |
| improper: |
sec; cc; mps |
 |
#3
 Plurals Plurals |
Unit symbols are unaltered in the plural. |
|
proper:
|
l = 75 cm |
|
improper:
|
l = 75 cms |
 |
#4
 Punctuation Punctuation |
Unit symbols are not followed by a period unless at the end of
a sentence. |
|
proper:
|
The length of the bar is 75 cm.
The bar is 75 cm long. |
|
improper:
|
The bar is 75 cm. long. |
 |
#5
 Multiplication Multiplication
& division |
A space or half-high dot is used to signify the multiplication of units.
A solidus (i.e., slash), horizontal line, or negative
exponent is used to signify the division of units. The solidus must not be repeated on
the same line unless parentheses are used. |
|
proper:
|
The speed of sound is about 344 m·s-1 (meters per second)
The decay rate of 113Cs is about 21 ms-1 (reciprocal milliseconds)
m/s, m·s-2, m·kg/(s3·A), m·kg·s-3·A-1
m/s, m s-2, m kg/(s3 A), m kg s-3 A-1 |
|
improper:
|
The speed of sound is about 344 ms-1 (reciprocal milliseconds)
The decay rate of 113Cs is about 21 m·s-1 (meters per second)
m ÷ s, m/s/s, m·kg/s3/A |
 |
#6
 Typeface Typeface |
Variables and quantity symbols are in italic type. Unit symbols
are in roman type. Numbers should generally be written in roman
type. These rules apply irrespective of the typeface used in the
surrounding text. For more details, see Typefaces for symbols in scientific manuscripts |
|
proper:
|
She exclaimed, "That dog weighs 10 kg!"
t = 3 s, where t is time and s is second
T = 22 K, where T is thermodynamic temperature, and K is kelvin |
|
improper:
|
He exclaimed, "That dog weighs 10 kg!
t = 3 s, where t is time and s is second
T = 22 K, where T is thermodynamic temperature, and K is kelvin |
 |
#7
 Typeface Typeface |
Superscripts and subscripts are in italic type if they represent
variables, quantities, or running numbers. They are in roman type
if they are descriptive. |
| subscript category |
 |
typeface |
 |
proper usage |
| quantity |
|
italic |
|
cp, specific heat capacity at constant pressure |
| descriptive |
|
roman |
|
mp, mass of a proton |
| running number |
|
italic |
|
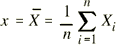 |
|
 |
#8
 Abbreviations Abbreviations |
The combinations of letters "ppm," "ppb," and "ppt," and the terms
part per million, part per billion, and part per trillion, and
the like, are not used to express the values of quantities. |
| proper: |
2.0 µL/L; 2.0 x 10-6 V;
4.3 nm/m; 4.3 x 10-9 l;
7 ps/s; 7 x 10-12 t,
where V, l, and t are the quantity symbols for volume, length, and time. |
| improper: |
"ppm," "ppb," and "ppt," and the terms part per million, part
per billion, and part per trillion, and the like |
 |
#9
 Unit Unit
modifications |
Unit symbols (or names) are not modified by the addition of subscripts
or other information. The following forms, for example, are used
instead. |
| proper: |
Vmax = 1000 V
a mass fraction of 10 % |
| improper: |
V= 1000 Vmax
10 % (m/m) or 10 % (by weight) |
 |
#10
 Percent Percent |
The symbol % is used to
represent simply the number 0.01. |
| proper: |
l1 = l2(1 + 0.2 %), or
D = 0.2 %,
where D is defined by the relation D = (l1 - l2)/l2. |
| improper: |
the length l1 exceeds the length l2 by 0.2 % |
 |
#11
 Information Information
& units |
Information is not mixed with unit symbols or names. |
| proper: |
the water content is 20 mL/kg |
| improper: |
20 mL H2O/ kg
20 mL of water/ kg |
 |
#12
 Math Math
notation
|
It is clear to which unit symbol a numerical value belongs and
which mathematical operation applies to the value of a quantity. |
| proper: |
35 cm x 48 cm
1 MHz to 10 MHz or (1 to 10) MHz
20 °C to 30 °C or (20 to 30) °C
123 g ± 2 g or (123 ± 2) g
70 % ± 5 % or (70 ± 5) %
240 x (1 ± 10 %) V
|
improper:
|
35 x 48 cm
1 MHz-10 MHz or 1 to 10 MHz
20 °C-30 °C or 20 to 30 °C
123 ± 2 g
70 ± 5 %
240 V ± 10 % (one cannot add 240 V and 10 %) |
 |
#13
 Unit Unit
symbols
& names
|
Unit symbols and unit names are not mixed and mathematical operations
are not applied to unit names. |
| proper: |
kg/m3, kg · m-3, or kilogram per cubic meter |
| improper: |
kilogram/m3, kg/cubic meter, kilogram/cubic meter, kg per m3, or kilogram per meter3. |
 |
#14
 Numerals & Numerals &
unit
symbols |
Values of quantities are expressed in acceptable units using Arabic
numerals and symbols for units. |
| proper: |
m = 5 kg
the current was 15 A |
| improper: |
m = five kilograms
m = five kg
the current was 15 amperes |
 |
#15
 Unit Unit
spacing |
There is a space between the numerical value and unit symbol,
even when the value is used in an adjectival sense, except in
the case of superscript units for plane angle. |
| proper: |
a 25 kg sphere
an angle of 2° 3' 4"
If the spelled-out name of a unit is used, the normal rules of
English apply: "a roll of 35-millimeter film." |
| improper: |
a 25-kg sphere
an angle of 2 ° 3 ' 4 " |
 |
#16
 Digit Digit
spacing |
The digits of numerical values having more than four digits on
either side of the decimal marker are separated into groups of
three using a thin, fixed space counting from both the left and
right of the decimal marker. Commas are not used to separate digits
into groups of three. |
|
proper:
|
15 739.012 53 |
|
improper:
|
15739.01253
15,739.012 53 |
 |
#17
 Quantity Quantity
equations |
Equations between quantities are used in preference to equations
between numerical values, and symbols representing numerical values
are different from symbols representing the corresponding quantities.
When a numerical-value equation is used, it is properly written
and the corresponding quantity equation is given where possible.
|
|
proper: |
(l/m) = 3.6-1
[v/(km/h)](t/s) |
| improper:
|
l = 3.6-1 vt,
accompanied by text saying,
"where l is in meters, v is in kilometers per hour, and
t is in seconds" |
 |
#18
 Standard Standard
symbols |
Standardized quantity symbols are used. Similarly, standardized
mathematical signs and symbols are used. More specifically, the
base of "log" in equations is specified when required by writing
loga x (meaning log to the base aof x), lb x (meaning log2 x), ln x (meaning loge x), or lg x (meaning log10 x). |
| proper: |
tan x
R for resistance
Ar for relative atomic mass |
| improper: |
tg x for tangent of x
words, acronyms, or ad hoc groups of letters |
 |
#19
 Weight vs. Weight vs.
mass |
When the word "weight" is used, the intended meaning is clear.
(In science and technology, weight is a force, for which the SI
unit is the newton; in commerce and everyday use, weight is usually
a synonym for mass, for which the SI unit is the kilogram.) |
 |
#20
 Quotient Quotient
quantity |
A quotient quantity is written explicitly. |
| proper: |
mass divided by volume |
| improper: |
mass per unit volume |
 |
#21
 Object & Object &
quantity |
An object and any quantity describing the object are distinguished.
(Note the difference between "surface" and "area," "body" and
"mass," "resistor" and "resistance," "coil" and "inductance.")
|
| proper: |
A body of mass 5 g |
| improper: |
A mass of 5 g |
 |
#22
 Obsolete Obsolete
Terms |
The obsolete terms normality, molarity, and molal and their symbols N, M, and m are not used. |
| proper: |
amount-of-substance concentration of B (more commonly
called concentration of B), and its symbol cB and SI unit mol/m3 (or a related acceptable unit)
molality of solute B, and its symbol bB or mB and SI unit mol/kg (or a related unit of the SI) |
| improper: |
normality and the symbol N, molarity and the symbol M
molal and the symbol m |
 |
