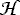 = 2/3
λ(3Sz2 -
S2) + γ(N
· S) + BN2
= 2/3
λ(3Sz2 -
S2) + γ(N
· S) + BN2In order to describe the rotational spectra of this class, Hund's coupling case (b) was chosen as the starting point. The rotational levels are characterized by the rotational angular momentum quantum number, N, and the resultant angular momentum quantum number, J, which includes the total electron spin angular momentum. If the molecule has nuclei with non-zero nuclear spin, I, these are coupled to J to form the total angular momentum quantum number F, whereby coupling case (bβJ) is assumed here. For pure case (bβJ) the electric dipole transitions occur with the selection rules: ΔN = � 1, ΔF = 0, � 1, and ΔJ = 0, � 1, in the absence of external fields. Since an intermediate coupling case is actually observed, transitions are allowed for ΔN = � 3. The magnetic dipole transitions occur with the selection rules: ΔN = 0, � 2 and ΔJ = 0, � 1.
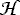 = 2/3
λ(3Sz2 -
S2) + γ(N
· S) + BN2
= 2/3
λ(3Sz2 -
S2) + γ(N
· S) + BN2
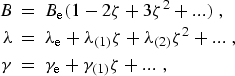 |
(eq10) |
where ![]() , the vibrational state
dependence of the molecular parameters is given by:
, the vibrational state
dependence of the molecular parameters is given by:
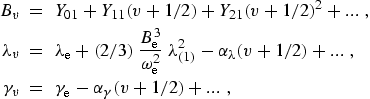 |
(eq11) |
where the Dunham coefficients, Ylj , are defined in section 2 and
| (eq12) |
| (eq13) |
The centrifugal distortion terms are defined as:
| (eq14) |
| (eq15) | |
| and | |
| (eq16) | |
With these definitions, the rotational energy levels are given in the form [9]:
| (eq17) |
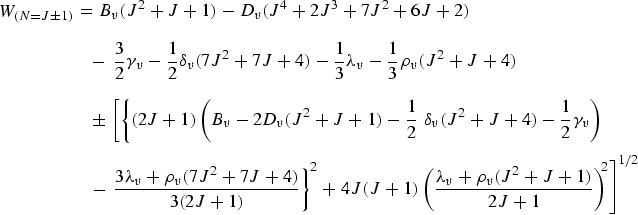 |
(eq18) |
The sextic terms, Hυ, of the rotational energy are neglected
because they cannot be determined from the data presently available for the
spectral observations on 3Σ
electronic ground state molecules. The energy equations are utilized with the
selection rules stated above to allow the determination of the molecular
constants
Bυ,
λυ, γυ, Dυ, ρυ,
and
Magnetic hyperfine structure has been described by Frosch and Foley [10] in terms of the determinable parameters, b and c. The nuclear electric quadrupole hyperfine structure is described by Amano, et al. [11] and results in determination of the constant, eQqυ, as defined in the discussion of 1Σ ground electronic state molecules.
| Symbols | (See section 2b for additional definitions.) | ||||||
| ai | Dunham potential coefficients. | ||||||
| λυ | Spin-spin coupling parameter in the υth vibrational state (MHz). | ||||||
| αλ | Spin-spin vibrational constant (MHz). | ||||||
| γυ | Spin-rotation coupling parameter in the υth vibrational state (MHz). | ||||||
| αγ | Coefficient in the power series expansion of γυ. | ||||||
| ρυ | Centrifugal distortion correction to λυ (MHz). | ||||||
| δυ | Centrifugal distortion correction to γυ (MHz) . | ||||||
| λe, λ(1), λ(2) | Expansion coefficients of λ in a power series of ξ. | ||||||
| γe, γ(1) | Expansion coefficients of γ in a power series of ξ. | ||||||
| b, c | Magnetic hyperfine coupling constants:
| ||||||
|
|||